
Hydroxypropyl Beta Cyclodextrin (HPBCD) CAS 128446-35-5 Pharmaceutical Cyclodextrin Derivatives
CAS Number: 128446-35-5
Product Category: Pharmaceutical Excipient
Main Function: Drug solubility enhancement and inclusion complex formation
Appearance: White or almost white amorphous powder
Water Solubility: Highly soluble in water
Applications: Drug formulation, injectable preparations, oral formulations, and topical drugs
Quality Standard: USP / EP / ChP Pharmaceutical Grade

Hydroxypropyl Beta Cyclodextrin (HPBCD) CAS 128446-35-5 Pharmaceutical Cyclodextrin Derivatives, also known as Hydroxypropyl Betadex, is a chemically modified cyclodextrin derivative widely used as a pharmaceutical excipient. It improves the solubility, stability, and bioavailability of poorly water-soluble active pharmaceutical ingredients (APIs), especially in injectable and oral formulations.
Item
Specification
Product Name
Hydroxypropyl Beta Cyclodextrin
CAS No.
128446-35-5
Synonyms
HPBCD, HP-β-CD
Type
Cyclodextrin Derivative
Grade
Pharmaceutical Grade
Function
Solubilizer, excipient
Hydroxypropyl Betadex is widely used in pharmaceutical applications including:
Used in parenteral formulations to improve solubility and prevent precipitation, ensuring safe and stable injectable solutions.
Enhances dissolution rate and bioavailability in tablets, capsules, and oral liquids, supporting consistent drug absorption.
Improves drug solubility in eye drop formulations while maintaining low irritation and high safety.
Facilitates rapid drug absorption through mucosal membranes in nasal sprays and inhalation systems.
Widely applied in veterinary formulations to improve drug performance and bioavailability.

With over 27 years of manufacturing experience, Xi’an Deli Biochemical Industry Co., Ltd. specializes in the production of cyclodextrin derivatives for pharmaceutical applications. Our core products include Hydroxypropyl Betadex (HPβCD), β-Cyclodextrin, and SBECD.
We supply high-quality excipients to pharmaceutical, veterinary, and chemical companies worldwide, supporting drug formulation, solubility enhancement, and advanced drug delivery systems.
Backed by stable production capacity and strong technical expertise, we provide consistent quality, regulatory support, and responsive service to meet the needs of global customers.

Hydroxypropyl Betadex is manufactured in accordance with major pharmacopoeial standards, including USP, EP, and ChP. Production is conducted under controlled conditions to ensure consistent quality and reproducibility across batches.
Quality control testing includes key parameters such as degree of substitution, identification, purity, and microbiological attributes, using validated analytical methods.
For detailed specifications and batch-specific data, including the Certificate of Analysis (COA), please contact us via the inquiry form.
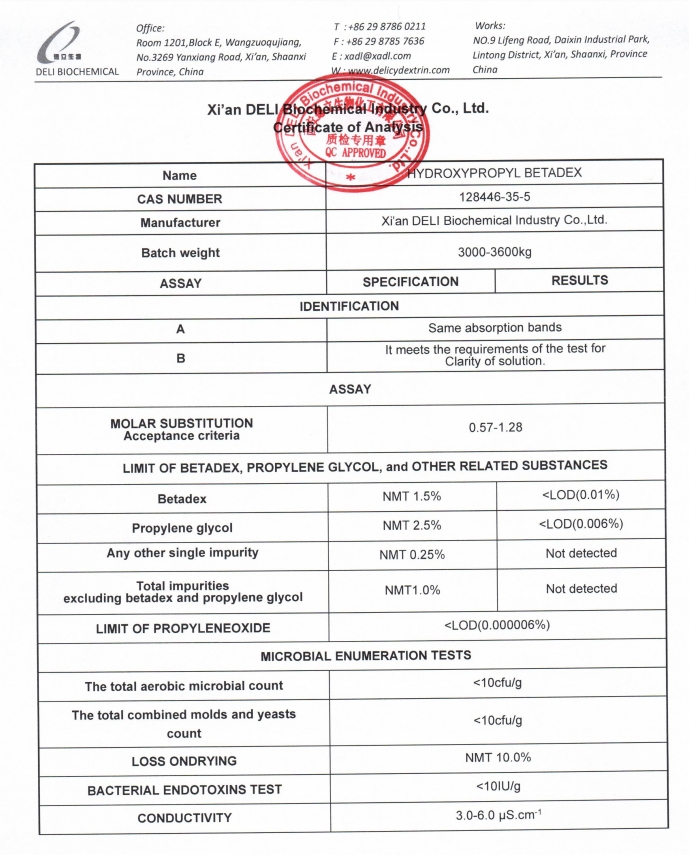
For full quality data and the latest COA, as well as other technical documentation, please contact us through our website Send Inquiry form. Our team will respond promptly.
✔ Extensive experience in cyclodextrin production
✔ Pharma grade quality meeting USP and EP standards
✔ Reliable supply with strong production capacity
✔ Dedicated technical support for formulation development
✔ Trusted by customers worldwide

We provide flexible ordering options and reliable international delivery to support our global customers.
36 months under recommended storage conditions
✔ T/T (Bank Transfer)
✔ L/C at sight
✔ Other payment terms can be discussed based on order volume and cooperation
✔ Express delivery (DHL, FedEx, UPS) for samples and small quantities
✔ Air freight for medium-volume orders
✔ Sea freight for bulk shipments
✔ Samples: typically available within 3–5 working days
✔ Bulk orders: depending on quantity and production schedule
We ensure safe packaging and reliable delivery to meet international shipping requirements.

Hydroxypropyl Beta Cyclodextrin (HPBCD) is a chemically modified cyclodextrin derivative used as a pharmaceutical excipient to improve the solubility and stability of poorly water-soluble drugs.
HPBCD is widely used as a solubilizer and excipient in pharmaceutical formulations, especially for injectable drugs, oral solutions, and drug delivery systems.
Yes, HPBCD is widely used in parenteral (injectable) formulations under pharmaceutical-grade standards, depending on regulatory approval and formulation design.
HPBCD is a modified derivative of Beta Cyclodextrin with improved water solubility and reduced toxicity, making it more suitable for pharmaceutical applications.
Yes, pharmaceutical grade Hydroxypropyl Betadex compliant with USP/EP standards is available upon request.
Yes, we provide full documentation support including COA, SDS, and Technical Data Sheet (TDS) for each batch.
The degree of substitution (DS) varies by specification and can be provided in the technical data sheet depending on customer requirements.
Yes, Hydroxypropyl Betadex is also widely used in veterinary pharmaceutical formulations to improve solubility and stability of active ingredients.
We are a professional manufacturer of cyclodextrin derivatives, including Hydroxypropyl Betadex (HPBCD), with our own production facility and quality control system, ensuring stable supply and consistent quality.
Yes, factory visits and third-party audits are welcome. We support customer audits to ensure transparency in our production process, quality system, and GMP compliance.