
Injection Grade Betadex Sulfobutyl Ether Sodium CAS 182410-00-0
Product Name: Betadex Sulfobutyl Ether Sodium
CAS No.: 182410-00-0
Grade: Injection Grade
Standard: USP / EP / ChP
Application: Pharmaceutical Excipient
Appearance: White to off-white, amorphous powder
Solubility: Freely soluble in water
Packaging: 500 g/bag, 1 kg/bag, 10 kg/drum; customized packaging available
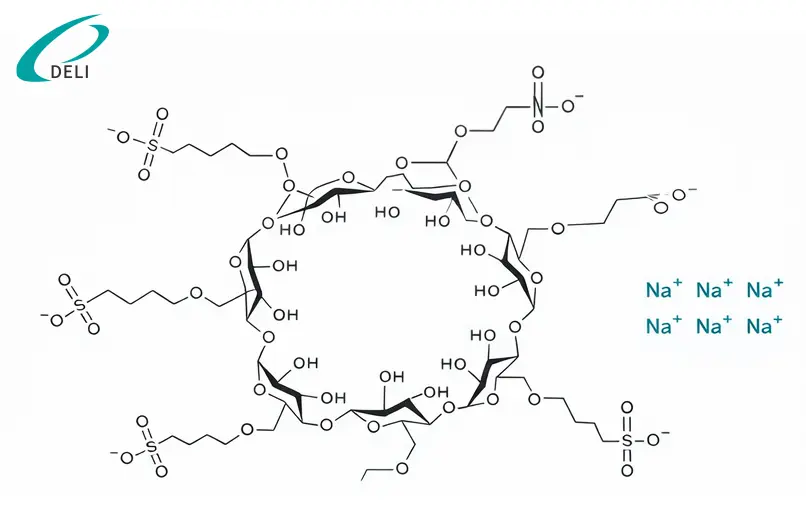
The product forms stable non-covalent inclusion complexes with active pharmaceutical ingredients, particularly basic and nitrogen-containing compounds. This interaction improves drug solubility and formulation performance without chemically modifying the API.
Betadex Sulfobutyl Ether Sodium Injection Grade is primarily used in parenteral and other non-oral dosage forms and is recognized for its favorable safety profile compared with native beta-cyclodextrin.
Product Name: Betadex Sulfobutyl Ether Sodium
CAS No.: 182410-00-0
Grade: Injection Grade
Standard: USP / EP / ChP
Application: Pharmaceutical Excipient
Appearance: White to off-white, amorphous powder
Solubility: Freely soluble in water
Packaging: 500 g/bag, 1 kg/bag, 10 kg/drum; customized packaging available
The product is manufactured under a strict quality management system covering raw material control, process validation, and finished product testing. Key quality attributes such as identification, assay, impurity profile, bacterial endotoxins, microbial limits, and degree of substitution are controlled according to pharmacopeial requirements.
Xi’an DELI Biochemical Industry Co., Ltd. holds complete corporate and production qualifications, including valid business licenses, pharmaceutical production permits, HALAL certification, DMF support, and other relevant regulatory documentation. Supporting technical and compliance documents are available upon request.

Injection Grade Betadex Sulfobutyl Ether Sodium is primarily applied in non-oral pharmaceutical dosage forms where enhanced solubility, stability, and safety are required. It is widely used in injectable solutions to improve the aqueous solubility of poorly water-soluble active pharmaceutical ingredients and to maintain formulation clarity and stability during storage and administration.
In lyophilized powders for injection, the product supports the formation of stable inclusion complexes, helping to protect sensitive APIs during freeze-drying and reconstitution processes. Its excellent compatibility makes it suitable for formulations requiring rapid dissolution and consistent performance after reconstitution.
The product is also applied in ophthalmic formulations, where low toxicity, high purity, and good tolerability are critical. By improving drug solubility and reducing irritation potential, it contributes to clear and stable eye drop solutions. In nasal preparations, it enhances drug dissolution and supports efficient delivery through the nasal mucosa while maintaining formulation safety.
In addition, Injection Grade Betadex Sulfobutyl Ether Sodium is suitable for inhalation formulations following appropriate formulation evaluation. Its ability to form reversible inclusion complexes and reduce the need for harsh solubilizers makes it a valuable excipient for advanced non-oral drug delivery systems.

The product provides excellent water solubility and strong solubilization capacity for poorly water-soluble active pharmaceutical ingredients, making it especially suitable for complex injectable formulations. Through the formation of stable and reversible non-covalent inclusion complexes, it significantly enhances formulation stability while maintaining the original chemical structure of the drug substance.
Compared with native beta-cyclodextrin and some conventional solubilizers, Betadex Sulfobutyl Ether Sodium exhibits a more favorable safety profile, with reduced hemolytic activity and lower renal toxicity, which supports its use in parenteral and other sensitive dosage forms. Its anionic structure contributes to improved compatibility with a wide range of APIs, particularly nitrogen-containing compounds.
In addition, the product supports flexible formulation development by improving drug bioavailability, reducing the need for organic solvents, and facilitating clear and stable aqueous solutions. This makes it a preferred excipient choice for modern injectable, ophthalmic, and advanced drug delivery systems.
Xi’an DELI Biochemical Industry Co., Ltd. has over 26 years of experience in the development and manufacture of cyclodextrins and their derivatives. Our long-term focus on pharmaceutical excipients allows us to deliver products with consistent quality, reliable performance, and stable supply for global customers.
We operate a dedicated production facility supported by a strict quality management system that covers raw material sourcing, in-process control, and finished product testing. This ensures batch-to-batch consistency and compliance with pharmacopeial requirements for injectable and non-oral pharmaceutical applications.
Our Betadex Sulfobutyl Ether Sodium Injection Grade is manufactured with controlled substitution and high purity, providing reliable solubilization and formulation stability for poorly water-soluble APIs. The product is designed to meet the technical and regulatory expectations of pharmaceutical formulation development.
DELI Biochemical holds complete corporate and manufacturing qualifications, including valid business licenses, pharmaceutical production permits, HALAL certification, and DMF support. Comprehensive technical documentation and regulatory assistance can be provided to support product registration and long-term cooperation.
With stable production capacity, professional technical support, and responsive communication, we are committed to building long-term partnerships based on quality, reliability, and trust.


Q: What is Injection Grade Betadex Sulfobutyl Ether Sodium used for
A: It is a pharmaceutical excipient used to improve the solubility and stability of poorly water-soluble active pharmaceutical ingredients through reversible inclusion complex formation.
Q: Is this product suitable for parenteral formulations
A: Yes. Injection Grade Betadex Sulfobutyl Ether Sodium is specifically designed for parenteral pharmaceutical applications, including injectable solutions and lyophilized powders for injection.
Q: Does this product chemically modify the API
A: No. It forms non-covalent inclusion complexes with APIs and does not alter the chemical structure of the drug substance.
Q: Which dosage forms can this product be used in
A: It is mainly used in non-oral dosage forms such as injectable solutions, freeze-dried injections, ophthalmic formulations, nasal preparations, and inhalation formulations, subject to formulation evaluation.
Q: How is this product different from native beta cyclodextrin
A: Compared with native beta cyclodextrin, Betadex Sulfobutyl Ether Sodium offers higher water solubility and a more favorable safety profile, making it suitable for injectable use.
Q: Is the product compliant with pharmacopeial standards
A: The product is manufactured under strict quality control and complies with pharmacopeial requirements such as USP and EP according to internal specifications.
Q: Are regulatory and technical documents available
A: Yes. Technical documentation, quality-related materials, and DMF support can be provided upon request to support formulation development and product registration.
Q: Does the manufacturer provide long-term and stable supply
A: Yes. Xi’an DELI Biochemical Industry Co., Ltd. operates dedicated production facilities to ensure stable supply and consistent batch-to-batch quality.
 Sulfobutyl Ether Beta Cyclodextrin Powder Injectable Grade Pharmaceutical Excipient
Sulfobutyl Ether Beta Cyclodextrin Powder Injectable Grade Pharmaceutical Excipient Betadex Sulfobutyl Ether Sodium CAS NO 182410-00-0
Betadex Sulfobutyl Ether Sodium CAS NO 182410-00-0 Sulfobutyl Ether Beta Cyclodextrin Sodium
Sulfobutyl Ether Beta Cyclodextrin Sodium Betadex Sulfobutyl Ether Sodium Injection Grade
Betadex Sulfobutyl Ether Sodium Injection Grade Betadex Sulfobutyl Ether Sodium SBECD Injectable Grade for Drug Formulations
Betadex Sulfobutyl Ether Sodium SBECD Injectable Grade for Drug Formulations Betadex Sulfobutyl Ether Sodium CAS 182410-00-0
Betadex Sulfobutyl Ether Sodium CAS 182410-00-0