
Hydroxypropyl Cyclodextrin HPBCD Pharma Grade USP EP
CAS Number: 128446-35-5
Product Category: Pharmaceutical Excipient
Appearance: White or almost white amorphous powder
Water Solubility: Highly soluble in water
Applications: Drug formulation, injectable preparations, oral formulations, and topical drugs
Quality Standard: USP / EP / ChP Pharmaceutical Grade
Hydroxypropyl Cyclodextrin (HPβCD) is a high-purity pharmaceutical excipient widely used to enhance the solubility, stability, and bioavailability of active pharmaceutical ingredients (APIs). It is commonly applied in pharmaceutical, cosmetic, and veterinary formulations.
As a modified β-cyclodextrin, HPβCD forms inclusion complexes with poorly soluble compounds, improving dissolution and enabling more effective drug delivery without altering the chemical structure of the API.
This pharma grade product complies with USP and EP standards, ensuring reliable quality and suitability for global pharmaceutical applications.
✔ Enhances drug solubility and bioavailability
✔ Improves stability of sensitive APIs
✔ Suitable for injectable, oral, and topical formulations
✔ Excellent safety profile and low toxicity
✔ Compatible with a wide range of pharmaceutical ingredients

Hydroxypropyl Cyclodextrin is widely used as a solubilizing and stabilizing excipient in modern drug delivery systems, supporting multiple dosage forms and administration routes.
HPβCD is used in parenteral formulations to improve solubility and prevent precipitation, ensuring stable and safe injectable solutions.
It enhances dissolution rate and improves bioavailability in tablets, capsules, and oral liquids.
HPβCD improves drug solubility in eye drops while maintaining low irritation and high safety.
Used in nasal sprays and inhalation systems to promote rapid absorption through mucosal membranes.
Widely applied in veterinary formulations to improve drug performance and absorption.
Xi'an Deli Biochemical Industry Co.,Ltd. Is a high-tech enterprise specializing in research & development, production & sales of cyclodextrin and its derivatives. Since its establishment on August 27, 1999,the company has been adhering to the quality Policy of "focusing on accessories, quality first, sincere service, striving for first-class". After more than 20 years of hard work and development, the company currently has products DELI Brand Hydroxypropyl Betadex, DELI Brand BetadexSulfobutyl Ether Sodium. The above products have been registered and filed with the
FDA.
We provide technical support and regulatory documentation including COA, DMF, and TDS to support global registration and compliance.
Our Hydroxypropyl Cyclodextrin meets the requirements of major pharmacopeias including USP, EP, and ChP. It is produced under a well-controlled manufacturing process to ensure consistent quality and reliable performance.
This product is suitable for formulation development, scale-up, and advanced drug delivery systems.
| Item | Specification |
|---|---|
| Appearance | White or almost white amorphous powder |
| Solubility | Freely soluble in water |
| pH (1% solution) | 5.0 – 7.5 |
| Degree of Substitution | 0.4 – 1.5 |
| Loss on Drying | ≤ 5.0% |
| Heavy Metals | Complies with USP/EP |
| Packaging | 500 g / 1 kg / 10 kg |
| Storage | Store in a cool and dry place |
| Shelf Life | 36 months |
A detailed Certificate of Analysis (COA) is available upon request.
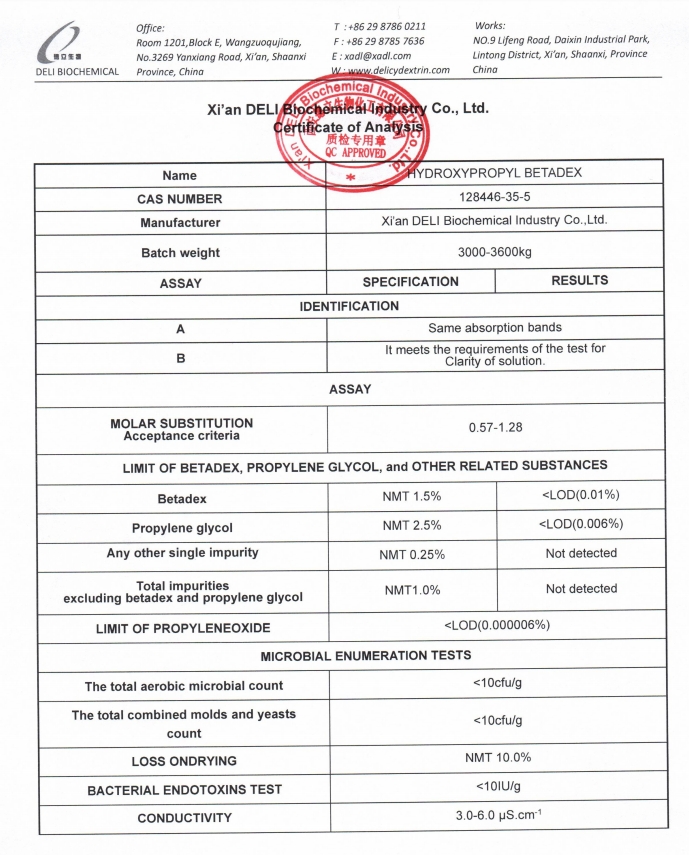
We provide high-quality Hydroxypropyl Cyclodextrin with reliable supply and strong regulatory support for global pharmaceutical companies.
✔ High purity and consistent quality
✔ Full regulatory support (DMF available)
✔ Free samples for evaluation
✔ Reliable supply and technical assistance
Our product helps customers accelerate formulation development and successfully enter international markets.


1. What is Hydroxypropyl Cyclodextrin used for?
It is used to improve solubility and bioavailability of poorly soluble drugs.
2. Is HPβCD suitable for injectable formulations?
Yes, it is widely used in injectable drug products.
3. Does it comply with USP/EP?
Yes, it meets major pharmacopeial standards.
4. Is DMF available?
Yes, DMF documentation is available upon request.
