
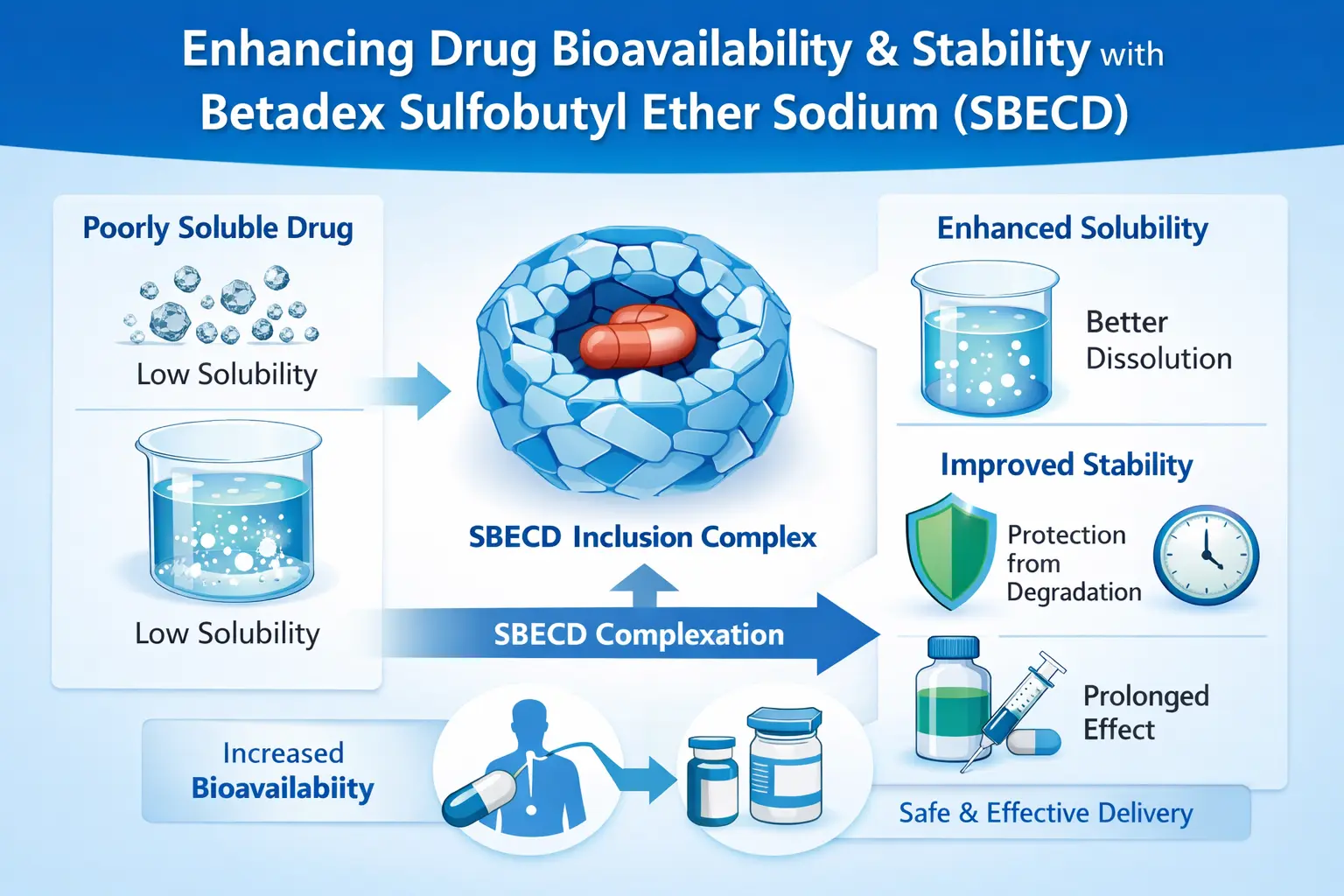
Betadex Sulfobutyl Ether Sodium (SBECD) changes the way medicines are made by combining with drugs that don't dissolve well. This cyclodextrin derivative improves bioavailability by encasing molecules, which makes some chemicals dissolve up to 1000 times better in water. The increase in stability happens because SBECD keeps drug molecules from breaking down while keeping their treatment effectiveness. As an FDA-approved excipient, SBECD makes it possible to administer medicines by mouth, which was not possible before. This is especially true for antifungal, antiviral, and oncology drugs.
What is bioavailability? It's the amount of a drug that gets into the body's systems without changing. Many potential pharmaceutical compounds have problems with bioavailability because they don't dissolve well in water. This makes it harder for the body to absorb them and use them effectively.
The unique chemical structure of SBECD helps it deal with this problem. The cyclodextrin backbone makes a space for drug molecules that is hydrophobic, and the sulfobutyl ether groups make it very easy for the drug to dissolve in water. Because it has two properties, SBECD is a great way to deliver drugs.
The creation of inclusion complexes happens naturally in water-based solutions. When drug molecules enter the cyclodextrin pocket, they form a stable but temporary bond. Since chemical bonds aren't used in this process, the drug's original structure and action are kept.
Researchers have found that SBECD complexation can make hydrophobic drugs seem to dissolve a lot better. The better solubility directly leads to better bioavailability, especially in parenteral forms where the drug needs to dissolve completely.
The best thing about this cyclodextrin derivative:
In the pharmaceutical business, Xi'an DELI Biochemical has more than 26 years of experience making cyclodextrin. Our factory can make more than 200 metric tonnes of goods every year, and batches are usually about 2.5 metric tonnes. This makes sure that there is a steady supply for large-scale activities.
DELI's quality control standards go above and beyond what the industry requires by using thorough testing methods. Each batch is carefully checked for the level of replacement, which makes sure that the complexation works at its best. Multiple methods are used in endotoxin testing to make sure that parenteral administration standards are met.
Our production process uses advanced cleaning methods to get rid of as few impurities as possible. The managed alkylation reaction makes sure that the substitution patterns are the same, which means that the behaviour of different batches can be predicted.
DELI is different from other suppliers because of how reliable their supply line is. Our well-established production lines and quality control systems help customers from the early stages of development all the way through commercial production. Long-term supply agreements give drug companies the security they need to start new products that do well.
As part of technical support, we can help with design and the creation of new analytical methods. Our expert team works with customers to find the best way to use SBECD in different situations, so that the benefits are maximised while costs and development time are kept to a minimum.

To use sulfobutyl ether cyclodextrin effectively, you need to carefully think about a number of formulation factors. The best SBECD concentration relies on the physicochemical properties of the drug and how much solubility improvement is wanted.
Phase solubility tests are very important for finding the right ratio of cyclodextrin to drug. These tests show how well the complexes work and help figure out what the lowest concentration of Betadex Sulfobutyl Ether Sodium is that will completely dissolve the drug.
Getting the right pH is a key part of making complexes as stable as possible. Even though SBECD can handle a wide range of pH levels, some drugs may have complexation behaviour that changes depending on the pH. Careful changes to the pH can make complex building and stability much better.
Temperature issues during preparation and storage can change the stability of a complex. Most SBECD complexes are stable at room temperature, but some versions last longer if they are stored in the fridge.
By checking for compatibility with other excipients, you can avoid interactions that might hurt the performance of the formulation. Preservatives, buffers, and antioxidants are some common medicinal excipients that work well with SBECD formulations.
Even though Betadex Sulfobutyl Ether Sodium has great safety ratings in clinical settings, it is still important to follow the right ways to handle and formulate it. In people, the excipient doesn't go through much metabolism; most of it is flushed out of the body unchanged by the kidneys.
Patients who are taking a high dose of SBECD or who are given it more than once may benefit from tracking their renal function. Nephrotoxicity is very rare with pharmaceutical-grade materials, but tracking gives you extra peace of mind.
The way a product is stored has a big effect on its quality and safety. SBECD should be kept in containers with tight lids that are kept away from water and too much heat. Keeping things in the right way keeps them from breaking down and keeps their performance traits stable.
Analytical testing is used to make sure that the quality and safety of a product are always the same during creation and production. Product integrity is maintained by testing it regularly for important factors such as degree of swap, impurity levels, and microbiological quality.
Betadex Sulfobutyl Ether Sodium is a big step forward in the science of pharmaceutical excipients. It makes it possible to formulate drug compounds that were hard to make before. Because it can improve stability and bioavailability at the same time, it is essential for current pharmaceutical development. The molecular encapsulation properties of SBECD are what make it work. They make drugs that don't dissolve easily into highly accessible forms. As drug companies keep making therapeutic compounds that are more complicated, SBECD gives them the formulation freedom they need to make products that work well during research and launch.
1. What is it about SBECD that makes it better than other solubilising agents?
SBECD has many special benefits, such as being very safe for living things, not being poisonous, being stable across a wide range of pH levels, and being able to create reversible inclusion complexes. SBECD is great for parenteral uses because it doesn't break down blood cells or irritate tissues like surfactants or organic solvents do.
2. What effect does SBECD have on the pharmacokinetics of drugs?
SBECD usually raises bioavailability by making drugs more soluble without changing the elimination routes too much. Cyclodextrin is a drug carrier that lets the active chemical out when it mixes with biological fluids. This method keeps the drug's natural pharmacological properties while making the delivery better.
3. What quality standards should drug companies have for SBECD suppliers?
Good SBECD has to follow strict USP/EP rules for medicinal excipients, which include limits of ≤0.25 EU/mg for endotoxins, heavy metals, solvents left over, and microbiological contamination. Suppliers should keep up with cGMP production standards and give full certificates of analysis.
DELI Biochemical is one of the biggest companies that makes Betadex Sulfobutyl Ether Sodium. They make injectable-grade ingredients that meet the strictest medicinal standards. We are the best partner for developing new drugs because we care about quality, dependability, and customer satisfaction. You can get the DELI advantage through our full technical help and guaranteed supply reliability. Contact us at xadl@xadl.com to discuss your specific formulation needs.

1. Stella, V.J., & He, Q. (2008). Cyclodextrins and their applications in pharmaceutical formulations: safety issues and toxicity considerations.Journal of Pharmaceutical Sciences, 97(5), 1585-1595.
2. Zia, V., Rajewski, R.A., & Stella, V.J. (2001). Effect of cyclodextrin charge on complexation of neutral and charged substrates: comparison of (SBE)7M-β-CD to HP-β-CD.Pharmaceutical Research, 18(5), 667-673.
3. Thompson, D.O. (1997). Cyclodextrins-enabling excipients: their present and future use in pharmaceuticals.Critical Reviews in Therapeutic Drug Carrier Systems, 14(1), 1-104.
4. Brewster, M.E., & Loftsson, T. (2007). Cyclodextrins as pharmaceutical solubilizers.Advanced Drug Delivery Reviews, 59(7), 645-666.
5. Rajewski, R.A., & Stella, V.J. (1996). Pharmaceutical applications of cyclodextrins: in vivo drug delivery.Journal of Pharmaceutical Sciences, 85(11), 1142-1169.
6. Luke, D.R., Tomaszewski, K., Damle, B., & Schlamm, H.T. (2010). Review of the basic and clinical pharmacology of sulfobutylether-β-cyclodextrin.Journal of Pharmaceutical Sciences, 99(8), 3291-3301.