
Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade CAS 182410 00 0 is supplied as a pharmaceutical excipient meeting USP and EP requirements. The product is manufactured as injection grade with controlled degree of substitution and consistent batch quality.
Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade is a highly water soluble anionic cyclodextrin derivative designed for pharmaceutical applications. Manufactured by Xi’an DELI Biochemical Industry Co., Ltd., this product is widely used as a pharmaceutical excipient to enhance the solubility, stability, and safety of poorly water soluble active pharmaceutical ingredients (APIs), especially in injectable formulations.
With controlled substitution degree and consistent quality, this injection grade product is suitable for regulated pharmaceutical development and commercial manufacturing.
Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade CAS 182410 00 0 is a chemically modified beta cyclodextrin with sulfobutyl ether groups. The anionic nature and high aqueous solubility allow it to form reversible inclusion complexes with a wide range of drug molecules through non-covalent interactions.
Compared with native beta cyclodextrin, SBE-β-CD demonstrates lower toxicity and improved compatibility for parenteral administration, making it one of the preferred cyclodextrin derivatives for injection grade pharmaceutical formulations.
Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade is mainly applied in non-oral pharmaceutical dosage forms, subject to formulation evaluation.
It is particularly suitable for drug candidates with low aqueous solubility and stability challenges during formulation development.
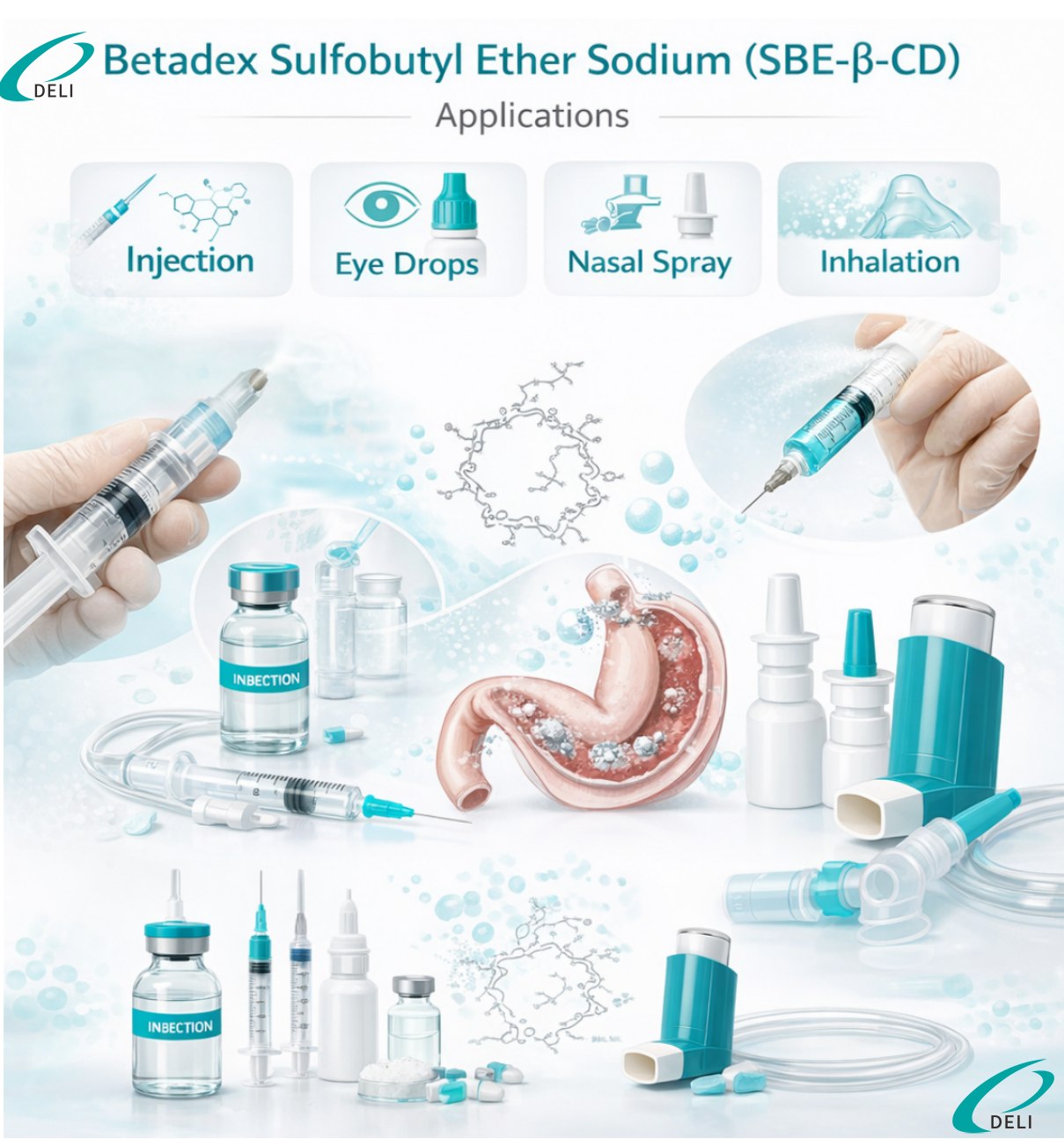
In injectable drug development, Sulfobutyl Ether Beta Cyclodextrin Sodium is used to improve drug dissolution, prevent precipitation after dilution, and enhance overall formulation stability. It also contributes to improved patient tolerance by reducing local irritation at the injection site.
The product is commonly selected for innovative drug delivery systems, complex injectable formulations, and drugs requiring high solubilization efficiency with proven safety.
Product Name: Sulfobutyl Ether Beta Cyclodextrin Sodium
CAS No.: 182410-00-0
Molecular Formula: C42H70-nO35·(C4H8SO3Na)n
Grade: Injection Grade
Executive Standard: USP / EP / Enterprise Standard
Application: Pharmaceutical Excipient
Packaging: 500 g/bag; 1 kg/bag; 10 kg/bag; 10 kg/drum
Storage: Store in a tightly closed container, protected from moisture and direct light.
This injection grade Sulfobutyl Ether Beta Cyclodextrin Sodium is manufactured under a comprehensive quality management system. Key quality attributes including identification, assay, average degree of substitution, residual impurities, endotoxin levels, and microbial limits are strictly controlled.
Each batch is tested to ensure compliance with pharmacopeial requirements and internal specifications, providing reliable performance for both formulation development and commercial pharmaceutical manufacturing.

A detailed Certificate of Analysis is provided for each production batch. The COA includes test items such as appearance, identification, assay, impurities, average degree of substitution, endotoxins, and microbial limits to ensure full traceability and quality transparency.

Q: Is this product suitable for injectable formulations?
A: Yes. This product is specifically manufactured as injection grade and is widely used in parenteral drug products.
Q: Does the product comply with USP and EP standards?
A: Yes. It is produced in accordance with USP and EP requirements as well as internal enterprise quality standards.
Q: Can samples be provided for formulation evaluation?
A: Yes. Samples can be provided upon request for formulation testing and technical evaluation.
Q: Is this product manufactured directly by your company?
A: Yes. Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade is manufactured in house by Xi’an DELI Biochemical Industry Co., Ltd. We are a direct manufacturer with our own production facilities and quality control system.
Q: Are you a manufacturer or a trading company?
A: We are a professional manufacturer specializing in cyclodextrin and cyclodextrin derivatives, supplying products directly from our factory without intermediaries.
Q: Is batch to batch consistency ensured?
A: Yes. Controlled production processes and strict quality control ensure consistent batch to batch performance.
Q: What dosage forms is this product typically used in?
A: It is mainly used in non oral dosage forms such as injectable solutions, lyophilized powders for injection, ophthalmic formulations, nasal preparations, and inhalation formulations, subject to formulation assessment.
Q: Is technical documentation available to support regulatory filing?
A: Yes. Supporting technical documents and quality related materials can be provided to assist formulation development and product registration.
Xi’an DELI Biochemical Industry Co., Ltd. has over 26 years of experience in cyclodextrin and cyclodextrin derivative manufacturing. The company focuses on pharmaceutical excipients and provides stable supply, consistent quality, and professional technical support to customers worldwide.
As a reliable manufacturer and supplier of Sulfobutyl Ether Beta Cyclodextrin Sodium Injection Grade, Xi’an DELI supports customers from early formulation development to commercial production.


 Sulfobutyl Ether Beta Cyclodextrin Powder Injectable Grade Pharmaceutical Excipient
Sulfobutyl Ether Beta Cyclodextrin Powder Injectable Grade Pharmaceutical Excipient Betadex Sulfobutyl Ether Sodium CAS NO 182410-00-0
Betadex Sulfobutyl Ether Sodium CAS NO 182410-00-0 Sulfobutyl Ether Beta Cyclodextrin Sodium
Sulfobutyl Ether Beta Cyclodextrin Sodium Betadex Sulfobutyl Ether Sodium Injection Grade
Betadex Sulfobutyl Ether Sodium Injection Grade Betadex Sulfobutyl Ether Sodium SBECD Injectable Grade for Drug Formulations
Betadex Sulfobutyl Ether Sodium SBECD Injectable Grade for Drug Formulations Betadex Sulfobutyl Ether Sodium CAS 182410-00-0
Betadex Sulfobutyl Ether Sodium CAS 182410-00-0